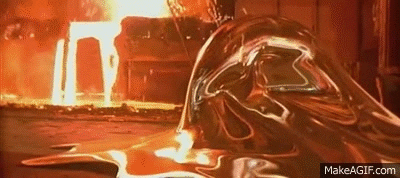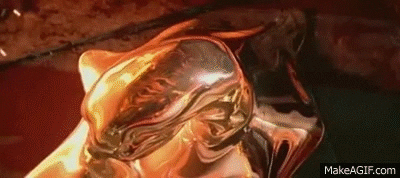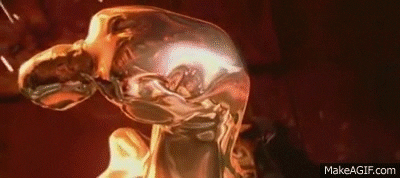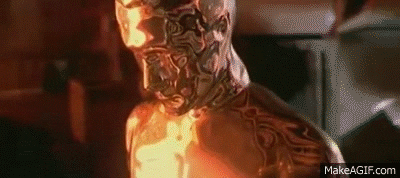Originally published at: http://boingboing.net/2017/06/14/all-about-gallium-the-metal-t.html
…
Been meaning to get some to play with, er… Let my kids play with? Yeah, it’s for the kids. With appropriate supervision of course.
Seeing this just reminded me, so I ordered. Wheeeee!
cool stuff but now you know if the reason why if you end up having kids w/ two heads.
Just don’t leave it in the scratched up bed of your F-150 on a hot day. Look up gallium induced structural failure on YouTube.
Liquid metals typically LOVE to mess with other metals if they can get through any surface coatings or native oxides. Even on Al it helps to disrupt the nm-ish thick Al2O3 to get anything dramatic.
Liquid Ga is really interesting.
It has one of the largest spans between its melting and boiling points at 1 atm.
- The crazy low vapor pressure makes it useful as a coolant.
Like Bi, Sb (I think), H2O and Pu the solid is less dense than the liquid. - This is kind of abnormal and also is why it’s packed in plastic and not glass.
Ga/In alloys are good replacements for Hg in thermometers.
Clean Ga on clean glass makes a good, though shortlived, mirror. It really wets glass.
And there’s a bottle of Ga sitting on my desk as I type this…
Theres better information about Ga in this thread than there is in all of that guys youtube channel.
I recollect reading a story about some lab that, as a practical joke, would give visitors gallium spoons with which to stir their tea. That’s a pretty good prank for someone who is not familiar with the metal. (The liquid metal puddles in the bottom of the cup, making it easy to avoid drinking it, and apparently pure gallium is not toxic. So if they managed to drink it anyways, the worst that happened is that the lab lost some gallium.)
I might be concerned that all the inexpensive gallium currently available on ebay is from China.
Thank you.
Oh, Ga is a good stabilizer of plutonium’s delta phase. Quite handy if you want to make something with the stuff. d-Pu is pretty awesome in its own way - it has a negative coefficent of thermal expansion!
Ironically, I think I missed that mention at Boingboing, but probably read a New Scientist review of that book… although I think I’ve also read of the pranks recounted elsewhere.
Isn’t there…uh…like…some metal that looks and acts a hell of a lot like this one, and people for centuries thought is was totally cool to use and even play with, and then one day someone thought “hmmm…I think this stuff might be making my miners sick so I better keep my mouth shut” but then eventually word got out and then eventually people considered it totally toxic? I’m not trying to say anything here, just pointing out some odd similarities that I keep coming back to.
We tried that with some early prototypes, but in vitro tests of proof-of-concept gallium-based mimetic pollyalloy embedded with rudimentary code functions just sat there and twitched.
@Slant, sounds like mercury a/k/a quicksilver. And it is totally toxic. The thing is liquid mercury isn’t very bioavailable, so that people could mostly get away with casual handling in the short term. But people who worked with it daily would get poisoned from chronic exposure to the vapour (c.f. the mad hatter).
Mercury salts are a different matter; some of those can kill with momentary skin contact.
As long as you aren’t playing with gallium halides or gallium salts you will be fine, unless you are one of the unlucky people who get dermatitis from prolonged exposure.
This topic was automatically closed after 5 days. New replies are no longer allowed.